Commercialisation and Outcomes

Real World Solutions (RWS)
Real World Solutions (RWS) generates real-world evidence to help clients achieve commercial success by addressing key strategic research questions for Payers, Patients, Providers & Regulators. Our industry-leading Real World Evidence (RWE) teams help to generate enhanced evidence for products throughout the development lifecycle by leveraging RWE expertise with specialised operational capabilities.
With our in-depth knowledge in the full scope of study designs ranging from secondary data to primary data collection (interventional & non-interventional), we provide evidence that shapes study feasibility and product development to determine the most efficient solutions.
Commercial Solutions Consulting
Our integrated Commercial Solutions Consulting team of 300+ subject matter experts within Insights, Evidence (Health Economics & Epidemiology, Pricing & Market Access, Patient Centred Outcomes, and Health Technology Assessment), Global Medical Communications and Creative & Digital Services provide strategic guidance and tactical solutions to define an asset’s path to market, optimise its value and generate evidence.
We plan for commercial success by reaching key stakeholders (regulators, payers, clinicians and patients) to gain approval, price, reimbursement, and adoption.

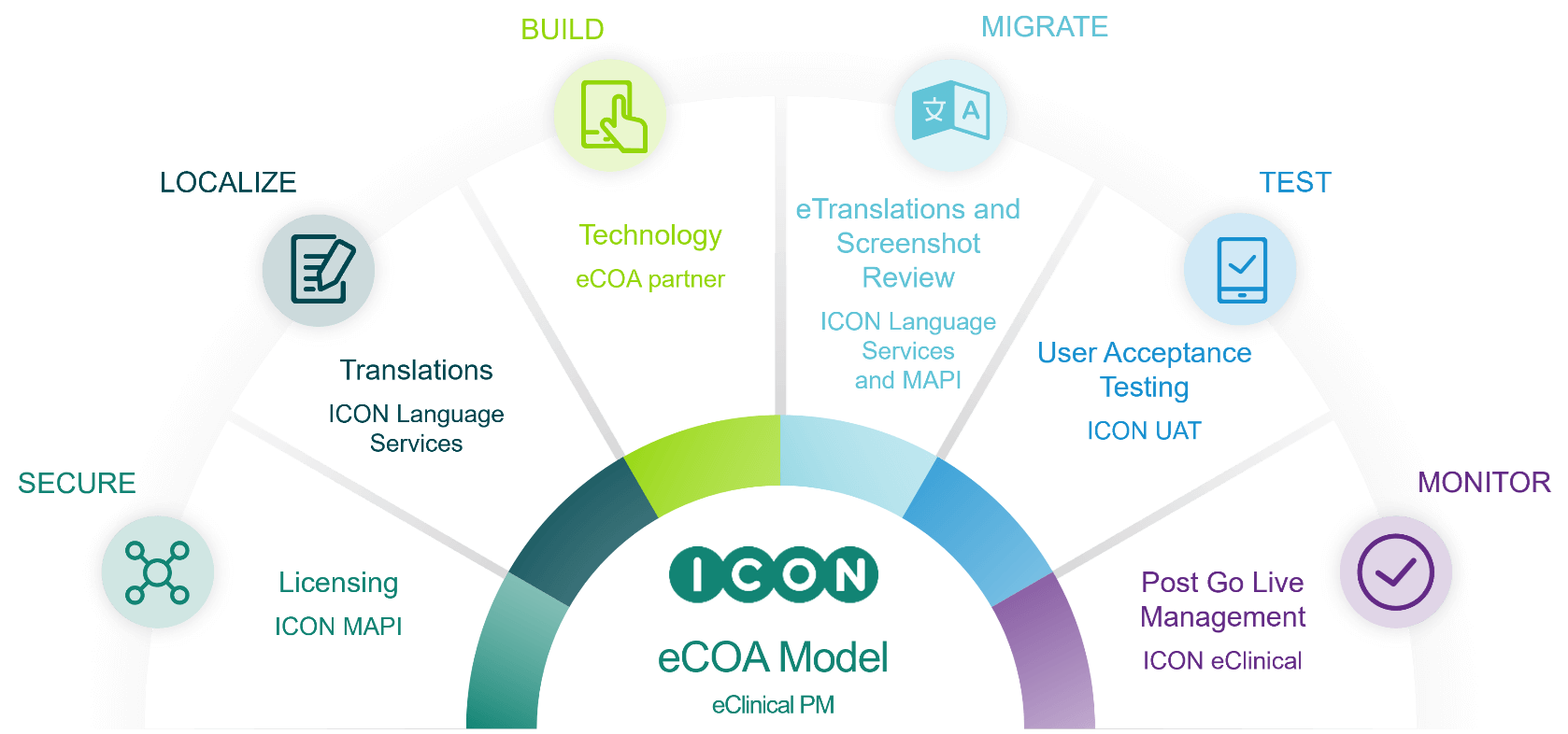
Patient Centered Sciences: Language Services & Mapi Research Trust
Language Services delivers specialised language solutions for clinical research and pharmaceutical/biotech content, and is the only ISO 17100, 9001 & 18587-certified, full-service language services provider within a top-tier CRO. Our professional linguistic resources perform high-quality translations and revisions of documents that are required in a wide variety of pharmaceutical- and clinical-related areas. Services include linguistic validation, voiceover, interpreting, ePRO migration and implementation, and transcription.
ICON is a leading provider of end-to-end Clinical Outcome Assessments (COAs) and eCOA solutions. As the primary sponsor of Mapi Research Trust, a non-profit organisation dedicated to improving patients’ quality of life by facilitating access to COAs for the entire scientific community, ICON presides over the industry’s largest database of COA information, and offers a single in-house solution for COA licensing, COA distribution, COA consultancies, eCOA validation and more.
Medical Device & Diagnostics Research
Our dedicated team at ICON can assist with all stages of development and testing by supporting the design, implementation and management of clinical trials for medical devices, combination products, in vitro diagnostics, digital therapeutics and software as a medical device throughout the development cycle. ICON’s Medical Device and Diagnostics Research group has over 30 years of global industry, product, technology and healthcare expertise.
Our medical device group is comprised of 130+ staff, dedicated to clinical operations, regulatory affairs, quality and strategy consulting. ICON’s medical device services are certified as conforming to the Quality Management System Requirements ISO 9001:2015.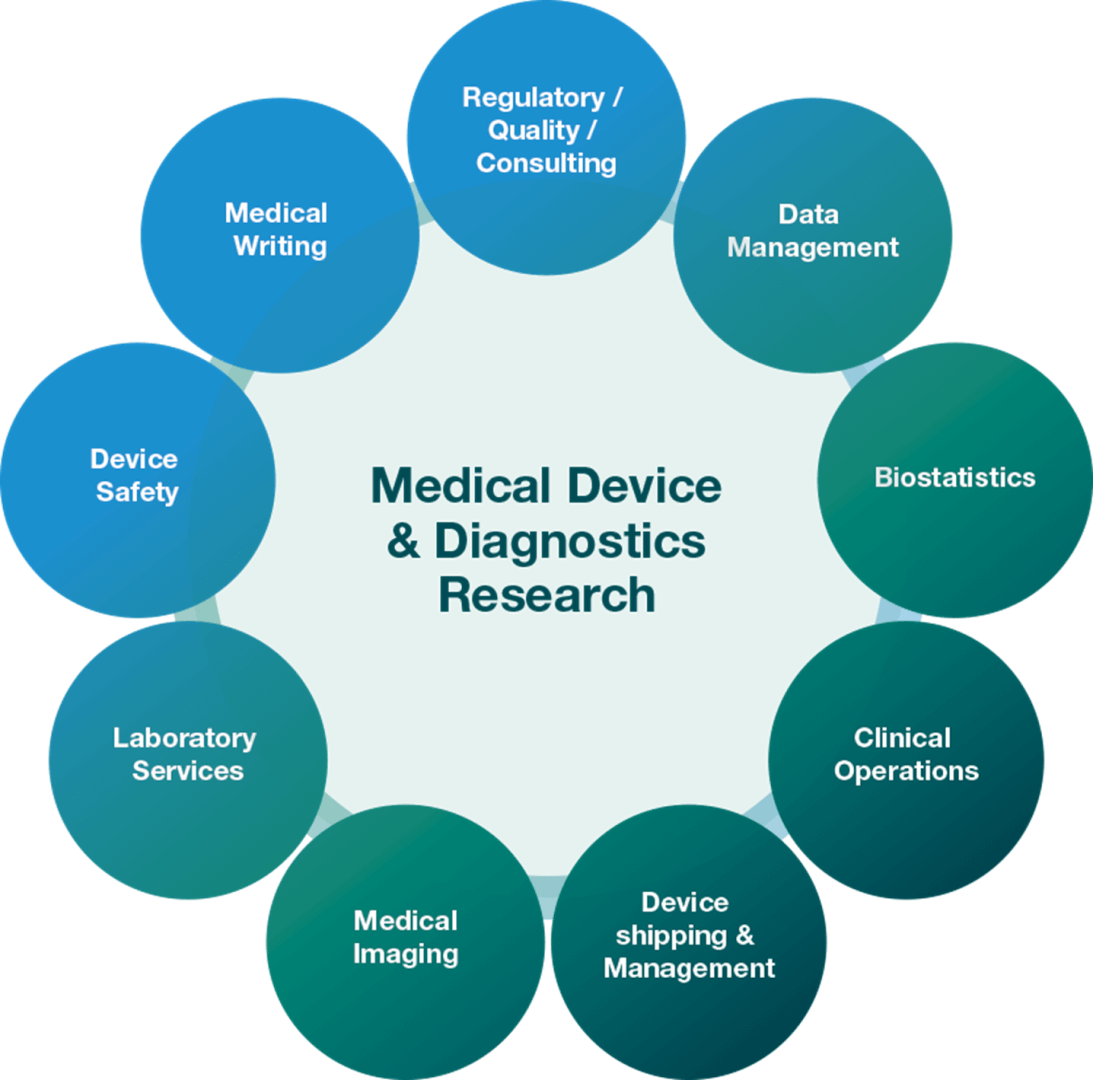
Roles in this area at ICON
Salary
Location
US, Blue Bell (ICON)
Department
Clinical Monitoring
Real World Solutions
Location
Los Angeles
Portland
San Antonio
Blue Bell
Long Beach
Remote Working
Remote or Office
Business Area
ICON Full Service & Corporate Support
Job Categories
Clinical Monitoring
Job Type
Permanent
Description
We are currently seeking a Senior Clinical Research Associate or Clinical Research Associate II to join our diverse and dynamic team. As a Clinical Research Associate at ICON, you will play a pivotal
Reference
JR147912
Expiry date
01/01/0001

Author
Kahla Cureton
Author
Kahla CuretonSalary
Location
Italy, Milan
Department
Clinical Monitoring
Real World Solutions
Location
Milan
Remote Working
Remote or Office
Business Area
ICON Full Service & Corporate Support
Job Categories
Clinical Monitoring
Job Type
Permanent
Description
We are currently seeking a Senior Clinical Research Associate (CRA) and CRA II's to join our diverse and dynamic ICON Biotech team. As a CRA at ICON, you will play a critical role in overseeing and ma
Reference
JR146456
Expiry date
01/01/0001

Author
Sophie Bingham
Author
Sophie BinghamSalary
Location
Netherlands, Utrecht, Zen
Department
Clinical Monitoring
Real World Solutions
Location
Utrecht
Remote Working
Remote or Office
Business Area
ICON Full Service & Corporate Support
Job Categories
Clinical Monitoring
Job Type
Permanent
Description
At ICON plc, we are a global healthcare intelligence and clinical research organization committed to driving innovation, excellence, and inclusion. Our mission is to advance clinical development and h
Reference
JR146303
Expiry date
01/01/0001

Author
Sophie Clarke
Author
Sophie ClarkeSalary
Location
Netherlands, Utrecht, Zen
Department
Clinical Monitoring
Real World Solutions
Location
Utrecht
Remote Working
Remote or Office
Business Area
ICON Full Service & Corporate Support
Job Categories
Clinical Monitoring
Job Type
Permanent
Description
At ICON plc, we are a global healthcare intelligence and clinical research organization committed to driving innovation, excellence, and inclusion. Our mission is to advance clinical development and h
Reference
JR145244
Expiry date
01/01/0001

Author
Sophie Clarke
Author
Sophie ClarkeSalary
Location
Regional United States (PRA)
Department
Clinical Monitoring
Real World Solutions
Location
Multiple US Locations
Remote Working
Remote or Office
Business Area
ICON Full Service & Corporate Support
Job Categories
Clinical Monitoring
Job Type
Permanent
Description
We are currently seeking a Clinical Research Associate II or Senior CRAs to join our diverse and dynamic team. As a Clinical Research Associate II at ICON, you will play a pivotal role in designing an
Reference
JR145857
Expiry date
01/01/0001

Author
Ana Lucia
Author
Ana LuciaSalary
Location
Poland, Warsaw
Department
Real World Solutions
Location
Sofia
Prague
Budapest
Warsaw
Bucharest
Remote Working
Remote or Office
Business Area
ICON Full Service & Corporate Support
Job Categories
Clinical Trial Support
Job Type
Permanent
Description
We are currently seeking a Site Management Associate II with French language to join our diverse and dynamic team. As a Site Management Associate II at ICON, you will play a vital role in supporting t
Reference
JR144475
Expiry date
01/01/0001

Author
Teodora Kaykieva
Author
Teodora KaykievaImpactful work. Meaningful careers. Quality rewards.
At ICON, our employees are our greatest strength. That’s why we are committed to empowering you to live your best life, both inside and outside of work. Whether your ambition is lead a global team, become a deep scientific or technical expert, work in-house with our customers or gain experience in a variety of different ICON functions, we will support you in realising your full potential. Learn more about Our Culture at ICON
